UA Scientist Identifies Cellular Gene Signatures for Heart Muscle Regeneration
A research team led by Jared Churko, PhD, director of the University of Arizona iPSC Core in the UA Sarver Heart Center, used a transcriptomic approach — studying what genes are expressed — to identify gene signatures of cell subpopulations identified as

Repairing heart muscle damaged by a heart attack or other cardiovascular diseases is one of the “holy grails” for cardiovascular scientists. The ability to repair heart muscle — especially by using a person’s own cells — would be a significant advance that could enhance quality of life for the millions of people who suffer from a heart attack or have a chronic heart condition.
Researchers believe that human-induced pluripotent stem cells (hiPSC) are the key to unlocking this regenerative ability. By taking a tiny bit of blood, scientists can generate an individual’s patient specific stem cells and then convert them into any cell type in the body — including cardiomyocytes, the cells that make up the heart muscle. The research, however, is in its infancy and the technique is not yet ready to be deployed for human heart disease regenerative purposes.

Jared Churko, PhD
Definitions:
- Transcriptomics is the study of the transcriptome — quantification of the types of RNA produced within the cell.
- Proteomics is the study of proteomes, the proteins expressed by a cell, tissue or organism.
- CRISPR gene editing is a technology for modifying an organism’s DNA code at the single-cell level. This has the potential to correct cells that are known to cause a heart condition.
The research reveals multiple subpopulations of cardiomyocytes expressing specific transcription factors (NR2F2, TBX5 and HEY2) — with different spatial and biological functions as observed in the heart. Dr. Churko believes this new understanding of cardiomyocytes can be used to better repair heart muscle injuries in the future.
“Understanding the gene signatures of different populations of hiPSC-CMs will impact our understanding of how to use such cells to discover drugs, model heart disease and repair a damaged heart,” Dr. Churko explained.
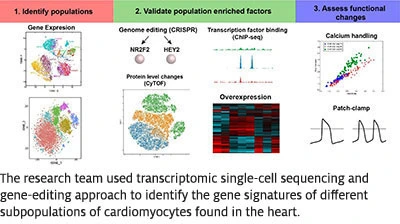
Please click here to view high resolution image of illustration.
Dr. Churko’s research team included scientists from Stanford University and the Cincinnati Children’s Hospital Medical Center. Dr. Churko is associated with the Center for Innovation in Brain Science, an assistant professor of physiological sciences and genetics in the UA Graduate Interdisciplinary Programs, member of the Center for Applied Genetics and Genomic Medicine and the UA BIO5 Institute and director of the UA iPSC Core in the UA Sarver Heart Center.
The Nature Communications article is titled, “Defining human cardiac transcription factor hierarchies using integrated single-cell heterogeneity analysis.” Co-authors include Joseph C. Wu, MD, PhD, Priyanka Garg, Haodi Wu, Jaecheol Lee, Quinton Wessells, Wen-Yi Chen, and Arun Sharma from Stanford Cardiovascular Institute. Other Stanford co-authors are Barbara Treutlein, Shih-Yu Chen, Gary Mantalas, Norma Neff, Garry Nolan and Eric Jabart.
Dr. Churko received funding from Canadian Institute of Health Research (201210MFE-289547), National Institutes of Health (1K99HL128906), and NIH Progenitor Cell Biology Consortium (PCBC_JS_2013/3_03). Dr. Wu received funding from NIH R01 HL126527, NIH R01 HL141371, NIH R01 HL130020, NIH R01 HL123968 and NIH HL128170.
Each year in the United States:
- About 610,000 people die of heart disease; that’s about 1 in 4 deaths.
- About 735,000 people have a heart attack.
- Coronary heart disease causes about 370,000 deaths.
- About 5.7 million adults have heart failure.
Source: U.S. Centers for Disease Control and Prevention
The University of Arizona Sarver Heart Center’s 150 members include faculty from cardiology, cardiothoracic surgery, pediatric cardiology, neurology, vascular surgery, radiology, endocrinology, emergency medicine, nursing, pharmacy and basic sciences. The UA Sarver Heart Center emphasizes a highly collaborative research environment, fostering innovative translational or “bench-to-bedside” research; dedicated to innovating lifesaving patient care. If you would like to give permission for Sarver Heart Center to contact you about heart research studies, please complete a Cardiology Research Registry Information Form. The academic mission of the Sarver Heart Center encompasses for fellowship programs in cardiovascular disease, interventional cardiology, advanced heart failure and transplant cardiology, and electrophysiology.

