Study shows how morphine may contribute to bone loss and cancer-induced bone pain
A study published in the journal PAIN is the first to show how morphine interacts with toll-like receptor 4 to contribute to pain evolution and bone loss.
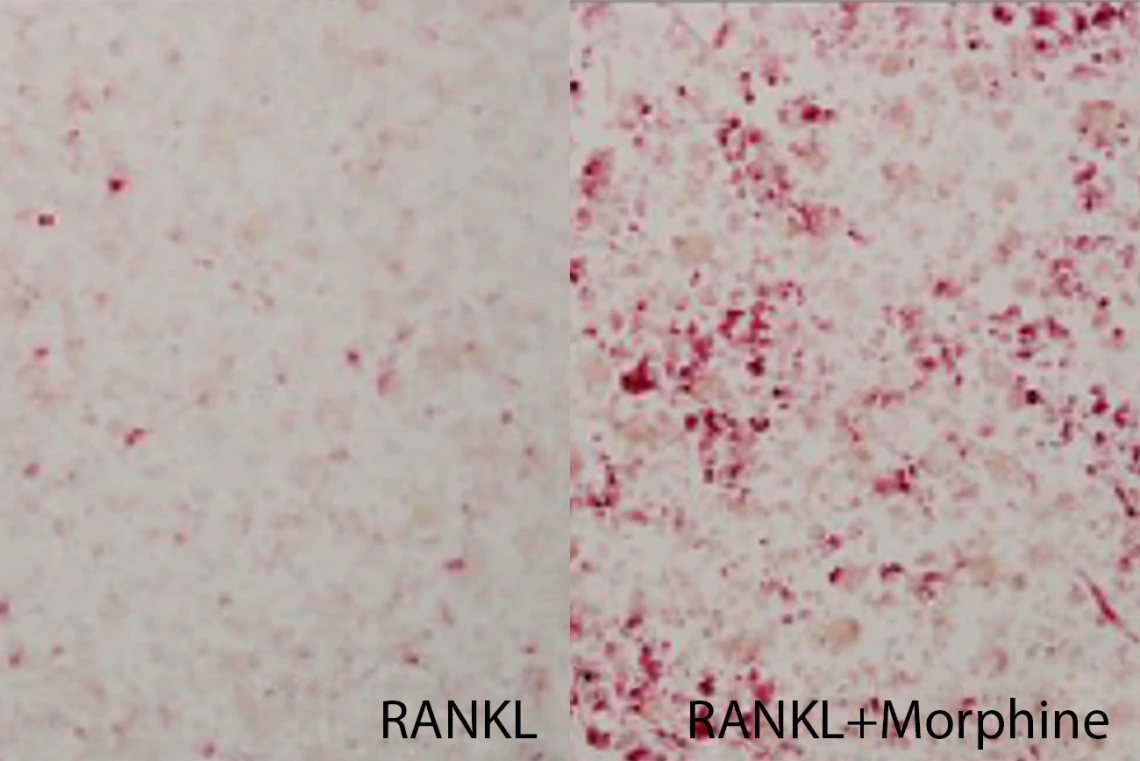
On the left, cells isolated from breast cancer cells were treated with RANKL, a growth factor that stimulates bone resorption, to produce osteoclasts (pink). When morphine was added to the cell culture (right), the number of osteoclasts increased, suggesting that morphine stimulated osteoclast production.
Opioids such as morphine are the gold standard for treating cancer-induced bone pain, but new University of Arizona Health Sciences research recently published in PAIN uncovered a mechanism by which chronic morphine use may increase bone loss and pain.
According to the National Institutes of Health, bone is the most common site for metastatic cancer, which is when cancer spreads from where it started to another part of the body. Nearly 70% of patients with prostate and breast cancers and about 30- 40% of patients with lung cancer develop bone metastases. The most frequent and important symptom of bone metastasis is pain.
“These findings suggest that chronic morphine for the treatment of things like metastatic cancer may not be the best since it may be actually decreasing bone density,” said senior author Todd Vanderah, PhD, director of the UArizona Health Sciences Comprehensive Pain and Addiction Center, professor and head of the Department of Pharmacology in the UArizona College of Medicine – Tucson and member of the BIO5 Institute and UArizona Cancer Center. “This does not mean doctors should remove opioids from their treatment plans, but it does highlight a need to develop alternative therapies that can lower the risk of side effects from opioids.”

Todd Vanderah, PhD, director of the UArizona Health Sciences Comprehensive Pain and Addiction Center, professor and head of the Department of Pharmacology in the UArizona College of Medicine – Tucson and member of the BIO5 Institute and UArizona Cancer Center.
Prior research in preclinical and human studies suggested opioids play a role in bone loss, with several studies tying chronic morphine use to lower bone density and increased time to hip fracture. This is the first study to identify a mechanism through which morphine contributes to increased bone loss and pain.
Dr. Vanderah and the research team examined how morphine interacted with mu opioid receptors and toll-like receptor 4, or TLR4, and compared the effects. Mu opioid receptors are the primary molecular targets for opioids and are responsible for the control of pain perception as well as side effects such as reward, tolerance, respiratory depression and others. Toll-like receptors recognize and bind to foreign molecules and are often found on immune cells, which typically increase in number when faced with an invader such as cancer.
In both laboratory and preclinical mouse models, researchers found that when morphine interacted with TLR4, it started a cellular cascade that included increases in inflammation and increased activity and production of osteoclasts, which are cells that degrade bone. The result was greater bone loss and increased pain. When TLR4 was blocked, there was no increase in pain or bone loss due to the morphine. The same effects were not seen when the mu opioid receptor was tested.
“We believe that morphine is acting through toll-like receptor 4 to increase the number of osteoclasts that are produced and the activity of those osteoclasts,” Dr. Vanderah said, explaining that as osteoclasts chew away more bone, pain levels rise. “In the bigger picture, chronic opiates may start enhancing bone loss in any inflammatory condition – if somebody has an activated immune system because of cancer or arthritis, for example.”
Additional research is needed, Dr. Vanderah said, to create a more detailed understanding of how opioids work not only at known opioid receptors, but also at off-target sites. Future findings could help predict the therapeutic pitfalls that arise in the treatment of cancer-induced bone pain and lead to better treatments for patients.
This study developed as an offshoot of one of Dr. Vanderah’s research projects in which cannabinoids were being examined for potential use as a non-opioid treatment for cancer-induced bone pain. That research is ongoing.
Co-authors on the study published in PAIN include: Tally Largent-Milnes, PhD, associate professor of pharmacology and member of the Comprehensive Pain and Addiction Center and BIO5 Institute; Jennifer Uhrlab, research scientist and laboratory manager in the College of Medicine – Tucson’s Department of Immunobiology; first author and former College of Medicine – Tucson MD/PhD student Austen Thompson, MD, PhD; College of Medicine – Tucson medical students Haley Ciccone and Dieter Mohty; and other former undergraduate and graduate students and research assistants.
This research was supported in part by the National Institutes of Health, the National Cancer Institute and the National Institute on Drug Abuse under award nos. R01CA14215 and P30DA051355.
Contact
Stacy Pigott
University of Arizona Health Sciences
520-539-4152
spigott@arizona.edu

