Aiming at a New Target to Stop a Widespread Common Virus
A postdoctoral researcher looks for ways to switch off the mechanism that allows a common yet potentially deadly virus to reproduce and thrive.

A Health Sciences strategic initiative is allowing postdoctoral researchers like Yeucheng Xi, PhD, to pursue work to create defenses against disease.
Yuecheng Xi, PhD, grew up wanting to be a pharmacist. But five years ago, after completing undergraduate degrees in pharmacology and toxicology, her father died from a herpesvirus he contracted after a kidney transplant. The experience changed Dr. Xi’s life.

Yuecheng Xi, PhD, with her father Hongliang Xi in 2010. His death was the inspiration for the work Dr. Xi is doing on human herpesvirus type 5, more commonly known as cytomegalovirus (CMV).
“I was looking for a lab that focused on this work and I found Dr. John Purdy's lab, which studies how the virus manipulates the host cells’ metabolism to replicate,” Xi added.
Since 2017, Dr. Xi has worked alongside John Purdy, PhD, assistant professor of immunobiology at the UArizona College of Medicine – Tucson, to understand how this common virus replicates. According to the Centers for Disease Control and Prevention, more than 50% of U.S. adults over 40 have CMV. It also is the leading cause of viral birth defects such as hearing loss, blindness, developmental disabilities and microcephaly (small head).
Creating defenses against disease
Dr. Purdy’s lab contributes to a larger UArizona Health Sciences strategic initiative called Personalized Defense, which addresses issues related to immunity, infection, inflammation and aging. Researchers in Dr. Purdy’s lab are zeroing in on complex metabolic interactions between viruses and host cells with the goal of better understanding of CMV biology to aid developing new antiviral therapies.

From left: John G. Purdy, PhD, assistant professor of immunobiology at the UArizona College of Medicine – Tucson, hopes to expand opportunities for postdoctoral researchers like Yeucheng Xi, PhD, with the support of the UArizona Health Sciences Personalized Defense strategic initiative.
“In normal human biology, cells generate many lipids to keep themselves happy, and the virus has evolved to take advantage of that process,” said Dr. Purdy, who also is a member of the BIO5 Institute and the Cancer Biology Program. “We want to uncover a molecule that will limit the ability of the cell to make the lipids the virus needs to set up a successful infection.”
In the lab, Dr. Xi purifies samples of CMV, adds them to cells, and tracks the process by which host cells make lipids and how the virus behaves during this interaction. She looks closely at the interactions that help viruses thrive.
In the short time Dr. Xi has been a part of Dr. Purdy’s lab, her work has already led to some answers.
Finding solutions, one step at a time
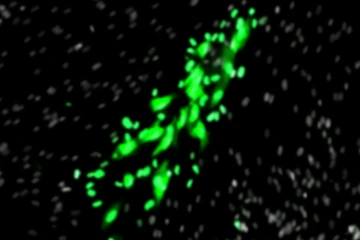
As part of Dr. Xi’s research, cells are infected by cytomegalovirus, shown in green florescence under a microscope.
Dr. Purdy says Dr. Xi has advanced the research by focusing on the mechanisms that viruses use to make new lipids and the stress response in host cells.
“What she has learned is the virus takes advantage of a stress response to reprogram lipid metabolism,” Dr. Purdy said. “Her work is uncovering what the virus is doing to the host to replicate and how the host cell is regulating lipid synthesis, learning not only about the virus, but about human biology.
“Her work aims to shut down the synthesis of some of these lipids. She aims to trick the cell into turning off certain processes to cut the virus off from what it needs,” he continued, adding that he hopes to expand opportunities for post-doctoral researchers with the support of the UArizona Health Sciences Personalized Defense strategic initiative.

Yuecheng Xi, PhD, earned pharmacology and toxicology degrees as an undergraduate before refocusing her career to understand viruses and help find therapies to combat viral infections.
“A virus can do very interesting things to help support its replication,” Dr. Xi said. “Lipids, overall, are induced upon infection. The most interesting thing is that several lipids with very long chain fatty acid tails did not exist in the host before and only show up upon infection. It seems that viruses need these very long chain fatty acids for some reason. We have some preliminary thoughts, but they are still not fully understood. Exploring the importance of these very long chain fatty acids to viral infection is critical for drug discovery as a potential target in the future.”
Our Experts
Contact
Margarita Bauzá
313-520-2109
mbauza@arizona.edu

