Testing a new therapeutic target for Alzheimer’s disease
Allopregnanolone is showing promise as a treatment for Alzheimer’s disease in elderly people and conditions including cerebral palsy in premature infants.
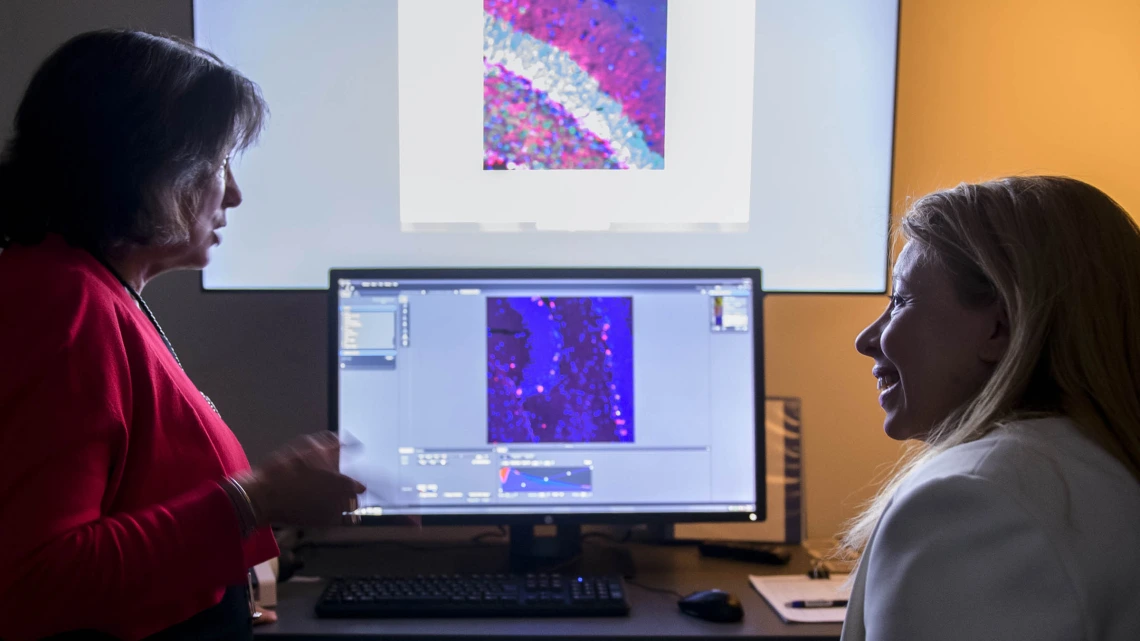
(From left) University of Arizona Health Sciences researchers Roberta Diaz Brinton, PhD, and Nahla Zaghloul, MD, are investigating the long-term safety and efficacy of allopregnanolone as a regenerative therapeutic for Alzheimer's disease and cerebral palsy.
One evening in the early 1990s, Roberta Diaz Brinton, PhD, treated nerve cells with allopregnanolone, a naturally occurring neurosteroid, and put them back in the incubator overnight. When she returned the next morning and peered through the microscope, she was amazed at what she saw.

Roberta Diaz Brinton, PhD, director of the Center for Innovation in Brain Science, is a leader in the field of Alzheimer’s, the aging female brain and regenerative therapeutics. She began mentoring Nahla Zaghloul, MD, when the two women discovered a common research interest in allopregnanolone.
“There were more nerve cells in the culture dish than there had been the evening before,” said Dr. Brinton, director of the Center for Innovation in Brain Science at the University of Arizona Health Sciences. “Our discovery was revolutionary because at the time, regeneration of nerve cells was not known. What we now know is that allopregnanolone is promoting the regeneration of neural stem cells within the brain. Our transformative discovery was the start of decades of research that has led to our upcoming clinical trial of allo as the first regenerative therapeutic for Alzheimer’s disease.”
Thanks to the work of Dr. Brinton and her team of researchers, allo is moving one step closer to becoming a new therapeutic option for people with Alzheimer’s disease. And, on the other side of the aging spectrum, it is showing promise for the treatment of cerebral palsy, which is normally diagnosed within the first few years of life.
Reversing the drivers of Alzheimer’s
Alzheimer’s disease is a progressive neurologic disorder that causes the brain to shrink and brain cells to die. It is an ultimately fatal condition that 6.5 million people in the United States currently live with, according to the Centers for Disease Control and Prevention. Symptoms usually begin after age 60.

More than 6 million Americans are living with Alzheimer’s disease, and deaths from Alzheimer’s have increased 145% from 2000 to 2019, according to the Alzheimer’s Association.
Dr. Brinton’s previous research found that allopregnanolone can restore the structural integrity and cognitive function of the brain during the early stages of the disease. It essentially combats or reverses many of the drivers of Alzheimer’s disease.
“We saw evidence for regeneration of the hippocampus, which is the area of the brain that is affected early in Alzheimer's disease,” said Dr. Brinton, explaining that the hippocampus is found in the inner folds of the brain and is critical for both learning and memory.
“We also saw evidence of an increase in white matter, which is important for fast communication between nerve cells within the brain critical for learning and memory circuits,” she added.
Dr. Brinton’s research has shown that in addition to promoting regeneration, allo improves the brain’s energy system, which is critical for the energetic demands of cognitive function, while reducing inflammation in the brain and the hallmark pathologies of Alzheimer’s disease.

Nahla Zaghloul, MD, assistant professor of pediatrics, focuses her research on periventricular leukomalacia, a white matter injury affecting the premature infant’s brain that is commonly associated with cerebral palsy.
“We are very excited by these findings because with just one therapeutic we will be able to promote regeneration of the brain, improve energy production in the brain, and reduce inflammation and the pathology of Alzheimer’s. It is very exciting,” Dr. Brinton said.
Taking the research a step further, Dr. Brinton is now leading a proof-of-concept Phase 2b, multi-site clinical trial called ReGenBrain©. The trial is designed to investigate the long-term safety and efficacy of allo to function as a regenerative therapeutic to restore structural integrity and cognitive function of the brain in participants with mild Alzheimer's disease dementia.
Study participants include people ages 55 to 80 who carry the APOE e4 gene, which has been shown to increase the risk of developing Alzheimer’s, and have been diagnosed with probable Alzheimer’s disease. Participants will be randomly placed into one of two groups: the first group will receive treatment with allo for 12 months and the second group will receive a placebo. After 12 months, all participants in the placebo group will be transitioned to receive allopregnanolone for the remaining six months of the study.
Potential brain-boosting effects for preterm infants
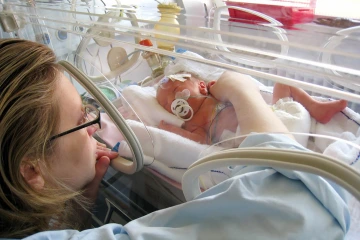
If successful, Allo could become the first regenerative therapeutic for Alzheimer’s and could also have a therapeutic impact on premature babies.
Allopregnanolone’s brain-boosting effects start before a person is born, during pregnancy. The third trimester is a time when the fetal brain undergoes a massive four-fold increase in size, and allo is seen at concentrations 25 times greater than earlier in pregnancy. Babies born preterm, especially those who miss the entire third trimester, do not receive the amount of allo they need. This could result in injury to the brain and the development of cerebral palsy.
“About half of children diagnosed with cerebral palsy were born prematurely,” said Nahla Zaghloul, MD, an assistant professor of pediatrics at the College of Medicine – Tucson. “There are a wide range of potential motor functions impacted, and the child’s mental capacity may also be impacted.”
Dr. Zaghloul, who collaborates and shares data with Dr. Brinton, is exploring how allopregnanolone could be used to help preterm infants.
“Our preliminary research shows the potential for much of the same anti-inflammatory, antioxidant, and neurogenerative effects that Dr. Brinton has seen in her research,” Dr. Zaghloul said.
Both researchers have found that a dose of allo that closely mimics the amount produced naturally in the third trimester yields the best results.
“Talk about Mother Nature getting the job done,” Dr. Brinton said. “She has done much of the work for us when it comes to allo being safe and effective. Now we are doing our part as scientists and clinicians to bring allo to patients who are counting on us.”
Our Experts
Roberta Diaz Brinton, PhD
Director, Center for Innovation in Brain Science
Professor, Pharmacology
Professor, Neurology
Professor, Evelyn F Mcknight Brain Institute
Professor, Psychology
Professor, Pharmacology and Toxicology
Professor, Clinical Translational Sciences
Member, BIO5 Institute
Nahla Zaghloul, MD
Assistant Professor, Pediatrics
Member, Steele Children’s Research Center
Attending Neonatologist, Banner Diamond Children’s Medical Center
Contact
Brian Brennan
Health Sciences Office of Communications
520-621-3510
brianbrennan@arizona.edu

