Examining the Microbiota’s Role in Infection, Immune Response
One postdoctoral researcher is focusing her work on the complex interactions between infection-causing pathogens and the bacteria that live in the body.

Kate Rhodes, PhD, is studying the interactions between the female reproductive tract microbiota and the bacterial pathogen that causes gonorrhea.
Many people hear about gonorrhea, one of the most common sexually transmitted infections in the United States, for the first time in a sex education class in school. They might know the infection can live silently inside the female reproductive tract, leading to complications like ectopic pregnancies, infertility and pelvic inflammatory disease.

(From left) Dr. Rhodes and mentor Magdalene So, PhD, use biochemistry, genomics and high-resolution microscopy to better understand the mechanisms of action of two closely related bacteria, <em>Neisseria gonorrhoeae</em> and <em>Neisseria meningitidis</em>.
“You have to figure out why things are happening the way they are and what is driving those mechanisms,” Dr. Rhodes said. “That has to happen before you can come up with a treatment later on.”
Understanding the environment
Bacteria live throughout the body, including in the female reproductive tract, and contribute to the microbiota, the system of microorganisms present in the human body. The living microorganisms that make up a healthy vaginal and cervical microbiota are not the same for every person, but an optimal community is characterized by having a large amount of Lactobacillus crispatus. Dr. Rhodes says the environment created by these bacteria can be the first line of defense against a pathogen like N. gonorrhoeae.
“It can be like a suit of armor,” she said. “It can be like another arm of the immune system in some instances.”

(From left) In Phoenix, Dr. Rhodes works with Melissa Herbst-Kralovetz, PhD, who has a long-standing interest and background in studying infections and diseases impacting women’s health. The Herbst-Kralovatz Lab has spent more than a decade working on the impact of different microbiota species on tissue environments.
Conversely, bacterial vaginosis is characterized by lactobacilli being depleted and replaced by different kinds of anaerobic bacteria. This environment may increase the risk of infection.
“On one side of the spectrum, you have low inflammation and a lactobacillus dominant community, and then on the other end, you have this really complex community and inflammation,” Dr. Rhodes said. “This environment could be setting the stage to be more predisposed to infection if you come into contact with things like gonorrhea, chlamydia or HIV.”
One of the premier labs to study N. gonorrhoeae and other bacteria belonging to the Neisseria genus is run by Dr. Rhodes’s mentor Magdalene So, PhD, in the College of Medicine-Tucson’s Department of Immunobiology.
“In the past, we studied N. gonorrhoeae in isolation because the techniques weren't there to do anything else,” said Dr. So, who is a member of the BIO5 Institute. “This project takes things further by examining the role of the microbiota and the immune response, as well as the interaction with harmful microbes.”
Researching the interaction
To better understand how N. gonorrhoeae reacts to the environment it enters, Dr. Rhodes is working with Melissa M. Herbst-Kralovetz, PhD, associate professor in the Department of Basic Medical Sciences and director of the Women’s Health Research Program at the UArizona College of Medicine-Phoenix.
In her lab, Dr. Herbst-Kralovetz pioneered the development of three-dimensional versions of the cellular tissue found in the organs of the female reproductive tract to study host-microbe interactions. She and her research team also grow and study the “good” bacteria that play an important role in the health of the system.
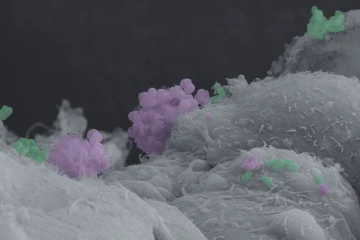
A microscopic image shows how <em>N. gonorrhoeae</em> (highlighted in pink) interacts with <em>A. vaginae</em> (highlighted in green), a bacteria associated with bacterial vaginosis.
“Dr. Rhodes works closely with a brilliant bacteriologist on my team,” Dr. Herbst-Kralovetz said. “She and Dr. Pawel Laniewski cultivate the bad and good bacteria in my lab and generate the 3D aggregates for her experiments.”
Dr. Herbst-Kralovetz’s work on the tissue model system and the impact of different microbiota species on those tissue environments spans more than a decade. The Herbst-Kralovetz Lab spends 28 days growing the tissue aggregates and preparing the environment before Drs. Rhodes and So can perform their experiments.
Dr. Rhodes bridges the gap between the two laboratories. She exposes the tissue and different microbiota to the pathogen, then transports the samples back to Dr. So’s lab for analysis.
“One of the things that Dr. Rhodes is looking at is the biogeography of how these bacteria are working in conjunction with one another – how these bacterial vaginosis-associated organisms are creating a biofilm, and then that interaction with N. gonorrhoeae,” said Dr. Herbst-Kralovetz, who is also a member of the BIO5 Institute. “We're able to then start to look at how does a key vaginal sexually transmitted infection pathogen react in the environment? Ultimately, we want to translate these findings to who is most susceptible to infection and experiencing poor outcomes.”
Dr. Rhodes uses bioinformatics to observe the behavior of N. gonorrhoeae as it struggles to find a niche in the healthy environment or finds an easy path to infection in the unhealthy environment. This could include looking at what genes are expressed and what proteins are produced.

Dr. Rhodes bridges the gap between the Herbst-Kralovetz Lab in Phoenix and the So Lab in Tucson by making regular trips across I-10.
“Iron acquisition is really important for bacteria in a human body. So, if we see all the genes for iron acquisition are turned on in one condition and not the other, then that makes us question maybe there is something to do with availability of iron sources in the host,” she said. “Maybe that differs in one condition or the other.”
As the three scientists search for ways to create better defenses against diseases such as gonorrhea, Dr. Herbst-Kralovetz points to the research as an exciting example of what can happen when people collaborate.
“We can learn from her, and she can learn from us,” Dr. Herbst-Kralovetz said of Dr. Rhodes. “It maximizes our resources, too, so she doesn’t have to try to repeat everything we have here in Phoenix down in Tucson. It has been incredibly valuable for all of us.”
Contact
Brian Brennan
Health Sciences Office of Communications
702-604-3088
brianbrennan@arizona.edu

